· Tea chemistry · 6 minutes
On the biochemical basis of oolong production. Jasmonic acid
Mechanization of sencha production in Japan began in the 1920s. By the 1960s, systems streamlining tea production were already quite commonly used. After initial preparation of the leaves, such systems allow for accelerating rolling, steaming, and drying of tea, among other processes.
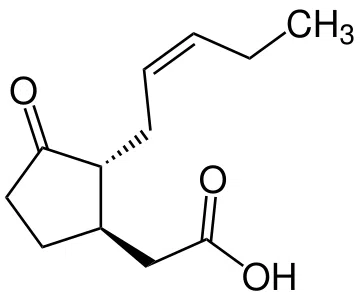
Many plants, including tea, respond to various adverse phenomena — mechanical damage, pathogens, drought, etc. — by increasing the production of volatile substances. Some of these substances directly serve protective functions — for example, they can repel pests or attract insects that feed on pests. The other part consists of signaling substances that trigger complex cascades of defensive reactions.
These volatile alcohols, aldehydes, ketones, etc. often have a pleasant aroma. Many of them contribute to the taste and aroma of tea, especially in the case of oolong and black teas. A number of technological operations in tea leaf processing aim to activate natural defense mechanisms by simulating certain stress factors. Weidiao — the wilting of tea raw materials — leads to moisture loss stress; sun wilting additionally provides exposure to elevated temperature and ultraviolet radiation; shaking — yao qing (搖青) — causes mild but prolonged and repeated mechanical damage; cooling — liang qing (晾青) — thermal stress. And all of this has little to do with polyphenol oxidation, to which various sources try to reduce the technology of oolong and black tea production.
Thus, for example, indole, a characteristic volatile component of oolongs, accumulates during the shaking of tea leaves, which is associated with increased expression of one of the subunits of tryptophan synthase (this enzyme catalyzes the formation of tryptophan from indole-3-phosphoglycerate and serine; this process occurs in two stages carried out by different subunits of the enzyme, and indole is an intermediate product). Interestingly, the destruction of cell walls during the crushing of tea leaves leads to impaired indole synthesis, which is why black teas contain relatively little indole [1].
Jasmine lactone and (E)-nerolidol also accumulate in oolongs during the process called zuo qing (做青), with shaking and cooling acting synergistically. One can only admire the diligence of the tea planters of the Qing era, who, knowing nothing about these substances, about the ways they are synthesized, about gene expression, established purely empirically that creating a complex and strong floral aroma requires precisely this combination of factors: careful shaking of the tea leaves and storing them at rest in a cool room.
Some volatile tea aromas are present in the leaves of tea plants in the form of glycosides — they are bound to carbohydrate residues. Their release under the influence of glycosidases during tea leaf processing is of great significance; the responsible enzymes are beta-primeverosidases and beta-glucosidases, and they have been thoroughly studied [2]. Note that this is not oxidation but hydrolysis — polyphenols and polyphenol oxidases have nothing to do with it! It turns out, however, that unlike the fermentation faxiao (发酵) of black tea, in zao qing hydrolysis is not activated — it seems that without significant destruction of cell walls, there is no contact between glycosidases and their substrates. Moreover, some glycoside aroma components actually accumulate instead! Let me repeat this once more: oolong processing is not a partial version of fermentation similar to black tea fermentation, but an entirely different process — in some aspects, as we can see, even the opposite one.
But how does the increased gene expression, increased enzymatic activity, and production of volatile aromatic substances occur? What is the signal that causes the rearrangement of metabolic pathways? After all, neither enzymes nor their genes can themselves “sense,” for example, mechanical damage.
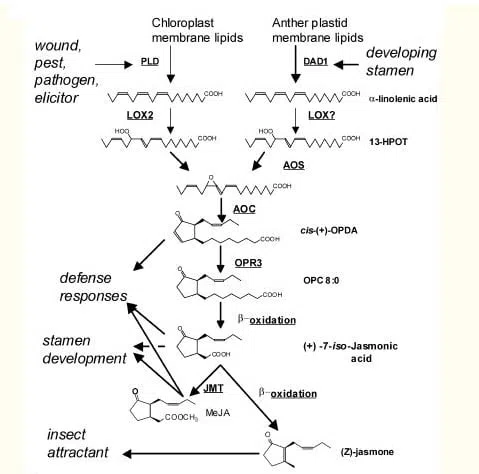
In plants, there are many substances that perform functions similar to hormones and mediators of inflammation and immune response in animal organisms. These include abscisic acid, salicylic acid, ethylene, etc., but in the case of oolong, the most interesting is jasmonic acid and its derivatives, as well as certain precursors, which are collectively called jasmonates.
Jasmonic acid regulates the processes of growth, aging and leaf abscission, root and bulb formation, and pollen development. But jasmonates also participate in plant responses to damage (for example, by an insect pest or herbivore) and the introduction of pathogenic bacteria and fungi. They trigger the synthesis of antimicrobial and insecticidal compounds, as well as proteins that disrupt the digestive process in insects. Both in terms of structure and function, they strongly resemble animal and human prostaglandins. Prostaglandins are formed from arachidonic acid, while jasmonic acid is formed from another polyunsaturated acid — alpha-linolenic acid. But if prostaglandins act locally, contributing to the isolation of the inflammatory focus (more precisely, inflammation is a mechanism for localizing the impact of a dangerous factor), then jasmonic acid spreads throughout the entire plant, providing a systemic response, and its ester — methyl jasmonate — is carried through the air to other plants, and they too begin to produce protective substances. Leafhoppers attack one bush — and the entire area of the tea plantation prepares for the attack.

Interestingly, despite their structural relationship with prostaglandins and leukotrienes, jasmonates in animal experiments display, on the contrary, anti-inflammatory properties. This is apparently due to their spatial configuration. Planar projections of jasmonate molecules are similar to inflammatory mediators, but their three-dimensional models largely overlap with non-steroidal anti-inflammatory drugs and can presumably bind to the same targets. Even greater interest is aroused by their anticancer potential: they have been found to be able to inhibit the growth of prostate cancer and induce apoptosis in lung cancer cells. For those who wish to study the jasmonate signaling pathway at a deeper level, I can recommend: [3]
In a study by scientists from the Tea Research Institute of the Guangdong Academy of Agricultural Sciences, it was established that as a result of shaking tea leaves, the content of jasmonic acid in them increases significantly, while the content of abscisic and salicylic acids does not change substantially (these are more responsible for responses to infection than to mechanical damage). Since some stages of jasmonic acid biosynthesis are associated with chloroplasts, the scientists compared the bleached shoots of an albino cultivar, whose cells have a reduced number of chloroplasts and disrupted structure (I wrote about these cultivars in the post about Anji Bai Cha), with green shoots of the same variety. It turned out that jasmonic acid production in bleached shoots decreases but not to zero. At the same time, the production of jasmine lactone, (Z)-3-hexen-1-ol, hex-1-enal, and hex-2-enal decreases — all these compounds are fatty acid derivatives, like jasmonic acid. A gene was also found whose expression increased after shaking only in green shoots. But a full deciphering of the mechanisms of tea leaf response to mechanical damage is still a long way off.
Activation of natural defense reactions through technological methods is a promising direction in tea production. Now that we at least partially understand their mechanisms, it is possible to devise ways to improve the aroma and taste of tea that ancient planters never stumbled upon. For example, in this study — [4] — scientists managed to influence the content of volatile substances by illuminating tea bushes with red light (wavelength 660 nm) and blue light (470 nm) before harvesting the leaves. Blue and red light increases the expression of 9/13-lipoxygenases, phenylalanine ammonia-lyase, and terpene synthases, which leads to increased synthesis of volatile fatty acid derivatives, phenylpropanoids, and terpenes, respectively.
Source: Tea House “Owl and Panda”



